Publications and Project Outcomes
This section highlights key publications, findings, and insights from the PICKED project, showcasing our contributions to chronic kidney disease (CKD) research and personalized medicine. Our work focuses on advancing biomarkers for early detection, developing models for disease progression, and exploring innovative intervention strategies to improve patient outcomes.
Our multidisciplinary team regularly publishes in high-impact journals and presents at international conferences, ensuring that our findings contribute to the broader scientific community. Stay tuned for the latest research updates and discoveries from the PICKED project!

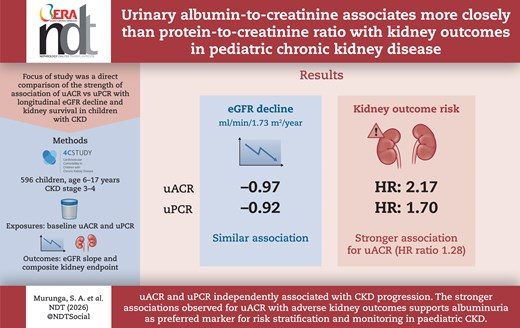
Paper spotlight: Albuminuria vs proteinuria in paediatric CKD, which predicts outcomes better?
Published in: Nephrology Dialysis Transplantation (NDT)
Authors: Sheila Murunga, Jonas Hofstetter (shared first authorship), et al. (4C Study Consortium)
Albuminuria vs proteinuria in paediatric CKD: a head-to-head comparison
In paediatric chronic kidney disease (CKD), both urinary albumin-to-creatinine ratio (uACR) and protein-to-creatinine ratio (uPCR) are established tools for risk stratification. But when it comes to predicting progression, it has not always been clear which marker carries the stronger prognostic signal.
In this new analysis from the 4C cohort (n=596 children aged 6–17 years with CKD stages 3–4), the authors directly compared uACR and uPCR in relation to eGFR decline and kidney survival outcomes.
Study overview
The study evaluated CKD progression using:
- annual eGFR slope, and
- a composite kidney outcome (50% eGFR loss, eGFR <10 mL/min/1.73m², or kidney replacement therapy).
Baseline uACR and uPCR were analysed in a harmonised way (log-transformed and standardised), enabling a true head-to-head comparison of effect sizes.
Key findings
🔹 Both markers matter
Both uACR and uPCR were independently associated with CKD progression in this paediatric cohort.
🔹 uACR shows a stronger prognostic signal for adverse kidney outcomes
Per 1 SD increase (log-transformed), the risk of the composite kidney outcome increased:
- 2.17× for uACR
- 1.70× for uPCR
Overall, uACR showed a ~28% stronger association with adverse kidney outcomes.
🔹 eGFR slope: no major overall difference
In the overall cohort, there was no clear relative difference between uACR and uPCR in their association with average eGFR slope change.
🔹 Differences become more apparent in advanced disease
The stronger uACR signal was particularly evident in children with advanced CKD and severe proteinuria.
Why this matters for personalised kidney care
This work reinforces a key message for paediatric nephrology and precision approaches in CKD:
Albuminuria is not only a marker of damage — it is a strong predictor of future outcomes.
By clarifying the relative prognostic strength of uACR vs uPCR, the study supports current guideline emphasis on albuminuria for:
- risk stratification, and
- monitoring intervention outcomes in paediatric CKD.
Read the full publication
Environmental and Genetic Determinants of Chronic Kidney Disease: New Insights from PICKED Researchers
Published in: Nephrology Dialysis Transplantation (NDT)
Authors: Maria Dolores Sánchez-Niño, Julia Byrska (PICKED Doctoral Candidate), Alberto Ortiz
Chronic kidney disease (CKD) affects more than 850 million people worldwide, yet its causes and risk factors remain complex. A newly published comment in Nephrology Dialysis Transplantation sheds light on how environmental exposures and genetic predisposition shape CKD development — and what this means for personalised kidney care.
Understanding CKD Risk: Beyond Genetics
Using extensive data from the UK Biobank, the authors explored the relative contributions of:
- Environmental exposures (“the exposome”)
- Age and sex
- Polygenic risk scores (genetic risk)
The exposome encompasses everything individuals experience throughout life, including lifestyle, pollution, socioeconomic status, housing conditions, smoking, and sleep habits.
Key Findings
🔹 Environmental factors outweigh genetics
For both CKD and cardiovascular disease, environmental exposures explain more variability in disease risk than genetic scores alone.
🔹 Everyday factors matter
Variables such as education, income, renting vs. owning a home, smoking behaviour, and even nap frequency showed strong associations with CKD risk.
🔹 Many exposures are actionable
Because many of these environmental factors can be changed or mitigated, the findings highlight new opportunities for prevention, early testing, and targeted public health interventions.
Implications for Personalised Medicine
This work reinforces a central message of PICKED:
True personalised kidney care must integrate biology, genetics, and the environments people live in.
Understanding how lived experiences shape disease risk moves us beyond a purely molecular view of CKD — toward a holistic approach where prevention and treatment are tailored to the individual.
Read the Full Publication

The exposome and incident CKD
Dietary Glycocalyx Mimetic Reduces Vascular Risk in Type 2 Diabetes
Published in: Diabetes Research and Clinical Practice (October 2025)
DOI: 10.1016/j.diabres.2025.112931
We are pleased to highlight a new publication by PICKED Doctoral Candidate Sajjad Biglari and collaborators from Mosaiques Diagnostics GmbH, reporting promising findings on the potential of dietary glycocalyx-mimetic supplementation to reduce vascular risk in individuals with Type 2 diabetes.
Study Overview
This study followed up on a previous placebo-controlled trial (NCT03889236) and examined the effects of an oral glycocalyx-mimetic supplement and a fasting-mimicking diet (FMD) on three established urinary peptidomic classifiers —
- HF2 (heart failure risk)
- CAD160 (coronary artery disease risk)
- CKD273 (chronic kidney disease risk)
The research involved 44 South-Asian Surinamese adults with Type 2 diabetes, who were monitored over a 12-week intervention. Urinary peptide profiles were analyzed using capillary electrophoresis–mass spectrometry (CE–MS), with validated support vector machine (SVM) classifiers used to assess molecular risk patterns.
Key Findings
Participants receiving the glycocalyx-mimetic supplement showed a significant reduction in HF2 risk scores (mean Δ = −0.58, 95% CI −0.83 to −0.33, p < 0.001).
Seventeen urinary peptides were significantly altered, mainly collagen-derived fragments, indicating improved extracellular-matrix turnover and potentially enhanced vascular health.
No meaningful effects were observed in the CAD160 or CKD273 classifiers, nor in the placebo or fasting-mimicking diet groups.
Conclusions
The study demonstrates that urinary peptidomics can sensitively detect early molecular responses to dietary interventions and highlights the potential of glycocalyx restoration strategies to lower cardiovascular risk in diabetes.
This work contributes to the growing evidence that molecular profiling through urinary peptidomics — one of the core methodologies employed within PICKED — can inform personalised prevention and therapy in chronic and metabolic diseases.
Authors: Sajjad Biglari, Lushun Yuan, Harald Mischak, Justyna Siwy, Agnieszka Latosinska, Miroslaw Banasik, Bernard M. van den Berg
Published: 25 October 2025

Urine sample collection flow-chart and study design outline. (A) Short representative flow chart of study protocol as performed previously of a subset of urine samples used in the present study and (B) Study design of endogenous peptidome research of a subset of urine samples.
Improving Haemodiafiltration through Smart Ultrafiltration Monitoring
We are excited to announce the latest publication from the PICKED project, led by RD Nephrologie SAS (RDN), now published in Scientific Reports under the title:
“In vitro evaluation of critical ultrafiltration fluxes and transmembrane pressure in a high flux dialyzer”
What’s the breakthrough?
High-volume haemodiafiltration (HDF) offers many benefits for patients with end-stage kidney disease. However, pushing for high convection volumes often creates practical complications — particularly pressure alarms and membrane clogging — which limit its use.
This new study evaluates the use of the GKD-UF max approach to define a critical and sustainable ultrafiltration rate. The research shows that:
- Stable and efficient ultrafiltration can be achieved by identifying the point of maximum filtration efficiency before fouling starts.
- The method helps prevent membrane clogging and reduces treatment interruptions, improving patient safety and dialysis quality.
- The approach can be implemented with existing online HDF machines, offering an accessible optimization path for dialysis centers.
Why is this important?
Haemodiafiltration is underused despite its benefits, partly because of the technical challenges it brings. This study offers a practical, data-driven solution to one of the key barriers: how to optimize ultrafiltration without causing damage or treatment failure.
Who contributed?
The research was conducted by Siavash Sohan Gir during his PhD at RDN within the PICKED MSCA Doctoral Network. It reflects the project’s mission to drive innovation in nephrology through interdisciplinary, patient-centered research and cutting-edge technology.
Acknowledgment
This work was supported by the European Union’s Horizon Europe Marie Skłodowska-Curie Actions (MSCA) under Grant Agreement No. 101168626 (PICKED project).
🔬 Stay tuned for more research updates from the PICKED network.
Follow us on LinkedIn, Bluesky and X for the latest news and publications!
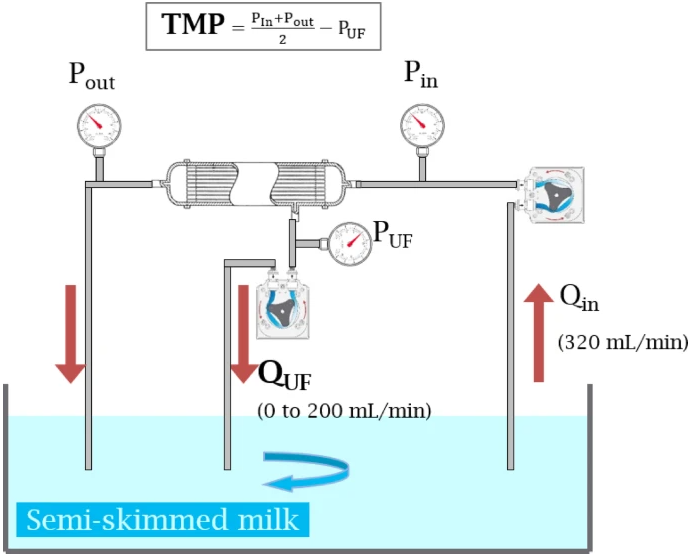
Diagram of the cross-flow filtration system used in all experiments.
Combination Therapy vs Biomarker-Guided Precision in Chronic Kidney Disease: Charting the Next Decade
Chronic kidney disease (CKD)—now affecting almost one billion people worldwide—demands therapies that can keep pace with its multi-factorial biology. In a new state-of-the-science review published in Biomolecules (Vol. 15, Article 809, June 2025), Sajjad Biglari, Harald Mischak and colleagues pose a decisive question: Should we rely on fixed-dose combination therapy (“polypill”) or move toward biomarker-guided personalised intervention?
Why this matters
CKD progression is driven by intersecting pathways—fibrosis, metabolic stress, haemodynamic overload and inflammation—so a single drug seldom suffices.
Modern “backbone” agents (SGLT2 inhibitors, non-steroidal MRAs, GLP-1 receptor agonists) already slow eGFR decline and curb cardiovascular risk.
Urinary-peptide classifiers (e.g., CKD273) now forecast renal or cardiac events years in advance, opening the door to precision escalation—or de-escalation—of therapy.
What the review highlights
Early combination regimens look powerful. Pilot trials that layer an SGLT2 inhibitor with finerenone (or add a GLP-1 RA) achieve > 50 % albuminuria reduction while mitigating hyperkalaemia risk. Yet cost and pill-burden remain hurdles.
Biomarker-guided treatment promises “right-patient-right-drug”. Risk classifiers can flag who is likely to benefit most from RAAS blockade, SGLT2i or MRA therapy, potentially sparing low-risk patients unnecessary exposure and expense.
A hybrid paradigm may win out. The authors envision a low-cost ARB + SGLT2i scaffold, fine-tuned by biomarker trends over time—a concept that dovetails with the PICKED network’s modelling work.
Read the full article
This research is supported by the PICKED project (HORIZON-MSCA-2023-DN-01, 101168626) and other European initiatives that are shaping the future of precision nephrology.

Summary of CKD progression, diagnosis, and respective treatment options.
Computational Drug Repositioning in Cardiorenal Disease: Advancing Therapeutic Innovation
Drug repositioning—the search for new therapeutic uses of existing drugs—has gained increasing momentum, particularly in tackling complex conditions like cardiorenal disease. While publications in this field continue to rise, the true measure of success lies in translating computational discoveries into real-world patient benefits.
We explore cutting-edge approaches in computational drug repositioning, leveraging bioinformatics, omics data integration, and network-based modeling to identify novel therapeutic opportunities. Despite significant progress, challenges remain in moving promising candidates from discovery to clinical application. Addressing these barriers with targeted strategies will be key to ensuring that repositioned drugs reach patients in need.
🔗 Read the full article here: DOI: 10.1002/pmic.202400109
This research is supported by the PICKED project (HORIZON-MSCA-2023-DN-01, 101168626) and other major European initiatives, driving forward the future of precision medicine.
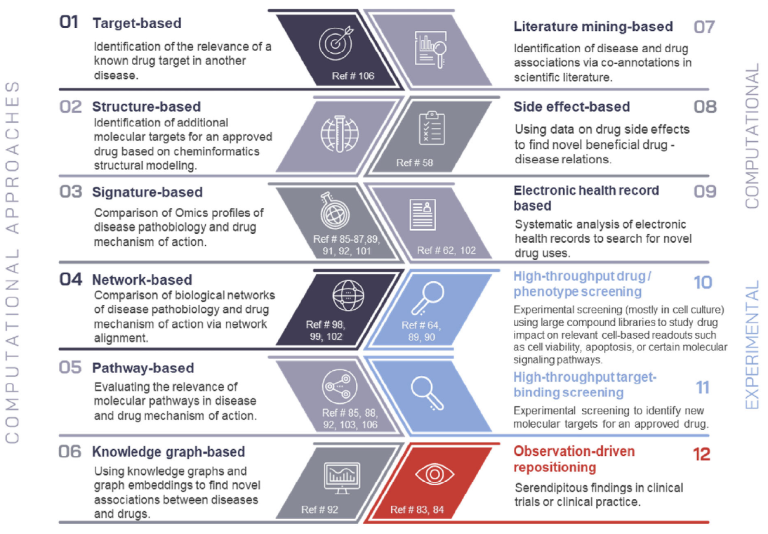
Overview on drug repositioning approaches.
Clinical Proteomics: From Scientific Breakthroughs to Patient Benefit
This article examines the progress and challenges of clinical proteomics, a field that has produced over 40,000 publications but has yet to make a widespread impact in clinical practice. Success should not be measured by publication volume alone but by tangible improvements in patient management and treatment.
We outline concrete solutions, emphasizing the need for a patient-centric approach to ensure that research efforts remain aligned with clinical utility. By addressing these barriers, we aim to shape the future of clinical proteomics, driving meaningful improvements in healthcare.
🔗 Read the full article here: DOI: 10.1002/pmic.202400346
This study represents a significant contribution to the field, supported by the PICKED project (HORIZON-MSCA-2023-DN-01, 101168626) and other major European research initiatives, driving innovation in personalized medicine.
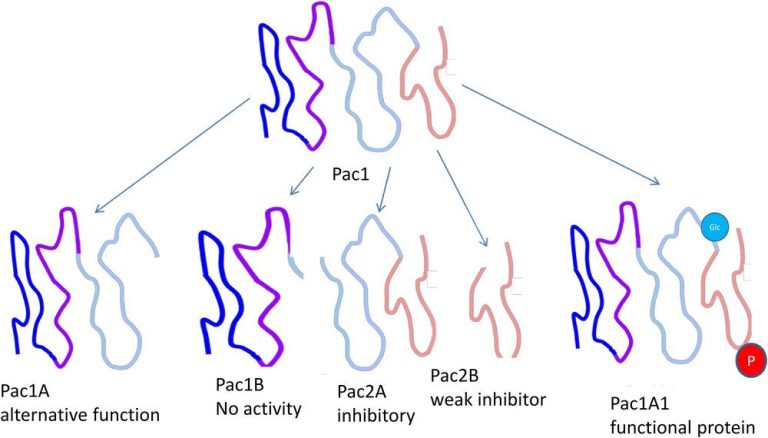
Biological functions of a hypothetical protein, Pac1, of which differently modified variants with different biological functions exist. The (hypothetical) protein Pac1 has little biological activity as a native polypeptide chain, but is converted into its active form, Pac1A1, by phosphorylation (red) and glycosylation (blue). The protein can also be specifically cleaved by proteases into Pac1A, which fulfils an alternative biological function, Pac1B which has no known function, and Pac2A and B, which inhibit the activity of Pac1A1.
Exploring the Role of GLP-1 RAs in CKD and Cardiometabolic Health
This article explores the potential of Glucagon-Like Peptide-1 Receptor Agonists (GLP-1 RAs) as transformative agents in the management of chronic kidney disease (CKD), type 2 diabetes (T2DM), obesity, and cardiovascular disease (CVD). These medications, including liraglutide, semaglutide, and tirzepatide, have shown significant benefits in metabolic control, weight reduction, and cardiovascular protection, making them an important consideration for nephrologists.
🔗 Read the full article here: https://pubmed.ncbi.nlm.nih.gov/39583142/
This marks an exciting milestone for the PICKED project, contributing valuable insights to the intersection of nephrology, metabolic health, and personalized medicine.
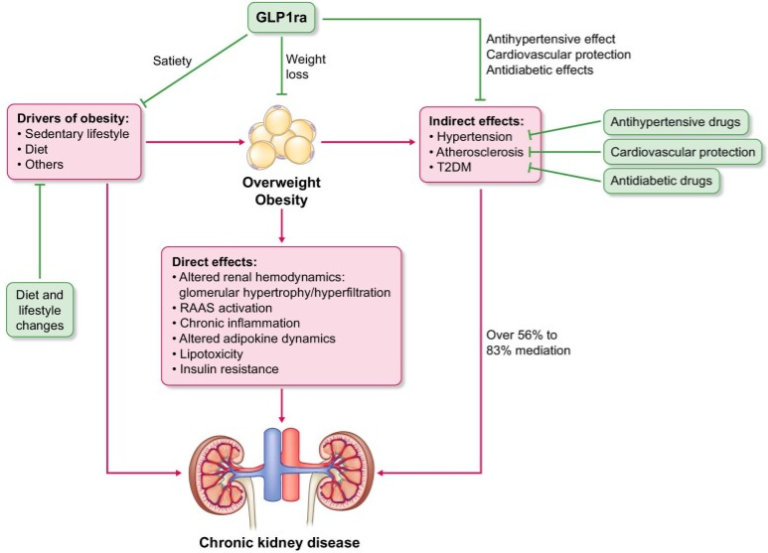
Obesity is an independent risk factor for CKD with a complex pathophysiology involving shared risk factors, direct effects of obesity and indirect effects through promotion of vascular disease, hypertension and T2DM. Indirect effects through BP and hyperglycaemia are estimated to mediate up to 83% of the negative impact of obesity on CKD. Mediation analyses of liraglutide and semaglutide in the T2DM CVOTs estimated up 48% mediation of kidney effects indirectly, through BP and hyperglycaemia. Similar analyses are awaited for overweight/obesity trials in which participants did not have T2DM.